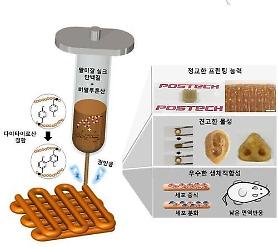
[Courtesy of T&R Biofab]
SEOUL -- HK inno.N, a pharmaceutical product maker in South Korea, will use 3D printed artificial skin to verify the efficacy of new drug substances for autoimmune disorder and skin diseases. Bioprinting can be used to print tissues and organs to help research drugs and pills.
HK inno.N, formerly known as CJ Healthcare, said it has signed a memorandum of understanding with T&R Biofab, a 3D bioprinting company capable of printing human organs and tissues for clinical transplantation. The two companies would expand cooperation and research on health functional foods and cosmetics materials.
"This collaboration with HK Inno.N will be a meaningful step toward full commercialization of our 3D printed skin and related drug screening technologies," T&R Biofab CEO Yoon Won-soo said in a statement. "We will complete innovative products that can be effectively used in industrial sites such as cosmetics and new drug development."
3D bioprinting combines cells, growth factors, and biomaterials to fabricate biomedical parts that maximally imitate natural tissue characteristics. The layer-by-layer method is used to deposit materials known as bioinks to create tissue-like structures. 3D bioprinting has begun to incorporate the printing of scaffolds that can be used to regenerate joints and ligaments.
Copyright ⓒ Aju Press All rights reserved.