[Gettyimages Bank]
Through a series of evaluations, the Ministry of Food and Drug Safety designates innovative medical devices with better capabilities than conventional equipment. The requirement needed for the commercialization of innovative devices is simplified to speed up the process.
The cornea is located on the outermost part of the eye to refract light and project image onto the retina which works in a similar way to a photographic film. Corneal disorders often occur among people who wear contact lenses or use smartphones for a long time. As the symptoms worsen, patients can experience eye inflammation which eventually causes vision to deteriorate.
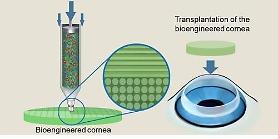
The drug safety ministry said it would continuously support the commercialization of artificial corneal transplants by revising various technical procedures such as designing clinical testing guidelines. "The device may be rolled out in 2023 after completing its clinical trial," a ministry researcher told Aju Business Daily.
Donated corneal transplants have been the only means of treating critical disorders before the ministry approved the new device. Patients had to wait for about eight years to find a donor.
Copyright ⓒ Aju Press All rights reserved.