Journalist

Gu Jun-hee
-
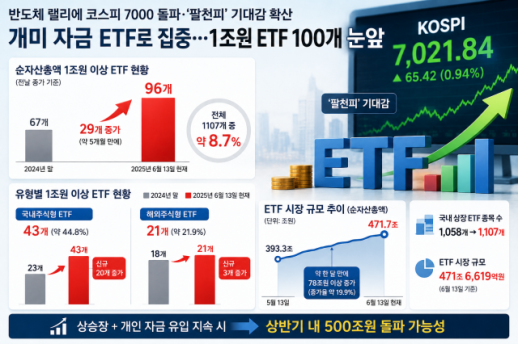 Retail Investors Shift Focus to ETFs Amid Market Surge As the domestic stock market continues to rise sharply, driven by semiconductor stocks, retail investor funds are rapidly flowing into the exchange-traded fund (ETF) market. Following the KOSPI's historic breach of the 7,000 mark, expectations for the '8,000 KOSPI' have grown, with nearly 100 ETFs surpassing 1 trillion won in total assets under management (AUM). According to the Korea Exchange on May 14, the number of ETFs with AUM exceeding 1 trillion won reached 96 as of the previous day's closing. This represents about 8.7% of the total 1,107 ETFs listed in South Korea. The number of ETFs exceeding 1 trillion won was only 67 at the end of last year, but it has increased by 29 in just five months, indicating rapid growth. Market analysts attribute this surge to the influx of retail investor funds into the ETF market amid the recent rise in the domestic stock market. The most notable growth has been seen in domestic equity ETFs. Of the 96 ETFs, 43 are focused on investing in companies listed on the Korean stock market, accounting for approximately 44.8% of the total. In contrast, overseas equity ETFs remain limited to around 21. At the end of last year, the number of ETFs with AUM over 1 trillion won was nearly equal between domestic (23) and overseas (18) equity ETFs. However, this year, as the domestic stock market rally centered on semiconductor and large technology stocks continues, 20 new domestic equity ETFs have surpassed the 1 trillion won mark. This shift is interpreted as a movement of investment funds towards the domestic ETF market due to the relatively superior returns of the Korean stock market compared to overseas markets. The overall size of the ETF market is also expanding rapidly. The number of listed ETFs has increased from 1,058 at the end of last year to 1,107 currently. The total AUM has surged as well, with the ETF market recently surpassing 450 trillion won and expanding to 471.66 trillion won as of the previous day. Notably, the total AUM of ETFs increased by over 78 trillion won in about a month, rising from 393.33 trillion won on April 13. This represents an increase rate of approximately 19.9%. The simultaneous strength of the stock market, particularly in the semiconductor sector, and the large influx of retail investor funds have accelerated the expansion of the ETF market. Industry experts suggest that if the current trend of fund inflows continues, the domestic ETF market could surpass 500 trillion won by the end of the first half of the year.* This article has been translated by AI. 2026-05-14 20:27:41
Retail Investors Shift Focus to ETFs Amid Market Surge As the domestic stock market continues to rise sharply, driven by semiconductor stocks, retail investor funds are rapidly flowing into the exchange-traded fund (ETF) market. Following the KOSPI's historic breach of the 7,000 mark, expectations for the '8,000 KOSPI' have grown, with nearly 100 ETFs surpassing 1 trillion won in total assets under management (AUM). According to the Korea Exchange on May 14, the number of ETFs with AUM exceeding 1 trillion won reached 96 as of the previous day's closing. This represents about 8.7% of the total 1,107 ETFs listed in South Korea. The number of ETFs exceeding 1 trillion won was only 67 at the end of last year, but it has increased by 29 in just five months, indicating rapid growth. Market analysts attribute this surge to the influx of retail investor funds into the ETF market amid the recent rise in the domestic stock market. The most notable growth has been seen in domestic equity ETFs. Of the 96 ETFs, 43 are focused on investing in companies listed on the Korean stock market, accounting for approximately 44.8% of the total. In contrast, overseas equity ETFs remain limited to around 21. At the end of last year, the number of ETFs with AUM over 1 trillion won was nearly equal between domestic (23) and overseas (18) equity ETFs. However, this year, as the domestic stock market rally centered on semiconductor and large technology stocks continues, 20 new domestic equity ETFs have surpassed the 1 trillion won mark. This shift is interpreted as a movement of investment funds towards the domestic ETF market due to the relatively superior returns of the Korean stock market compared to overseas markets. The overall size of the ETF market is also expanding rapidly. The number of listed ETFs has increased from 1,058 at the end of last year to 1,107 currently. The total AUM has surged as well, with the ETF market recently surpassing 450 trillion won and expanding to 471.66 trillion won as of the previous day. Notably, the total AUM of ETFs increased by over 78 trillion won in about a month, rising from 393.33 trillion won on April 13. This represents an increase rate of approximately 19.9%. The simultaneous strength of the stock market, particularly in the semiconductor sector, and the large influx of retail investor funds have accelerated the expansion of the ETF market. Industry experts suggest that if the current trend of fund inflows continues, the domestic ETF market could surpass 500 trillion won by the end of the first half of the year.* This article has been translated by AI. 2026-05-14 20:27:41 -
 Oh Se-hoon Proposes Debate with Jung Won-o Ahead of Local Elections Oh Se-hoon, the People Power Party candidate for Seoul mayor, reiterated his call for a one-on-one debate with Jung Won-o, the Democratic Party's candidate, on May 14, just six days before the local elections. Oh suggested that Democratic Party leader Jung Cheong-rae could moderate the debate, stating, "I hope at least one or two debates can be held." Speaking at a forum hosted by the Korean Newspaper and Broadcasting Editors Association in central Seoul, Oh said, "I am open to any format, time, place, or topic for the debate." He expressed frustration, saying, "Whenever I propose a debate, Jung gives evasive answers. Considering the public's right to know, I hope he reconsiders and shows at least a minimum level of courtesy." Oh emphasized the significance of the upcoming election, stating, "This election is a crossroads for whether the Lee Jae-myung administration will adopt a humble approach or transition into a more overt form of soft dictatorship. Absolute power corrupts absolutely; this is a historical truth." He warned that if Democratic Party candidates win in major local governments like Seoul and Busan, the current soft dictatorship under President Lee Jae-myung could become more severe. "I hope a minimum foothold for checking the government can be secured," he added. Oh also highlighted the choice between returning to the past of former Mayor Park Won-soon or moving toward the future. He pointed out that during Park's ten-year tenure, 389 redevelopment and reconstruction projects were canceled, leading to a significant reduction in housing supply and contributing to the current real estate crisis. He noted, "The people who implemented those policies are still within the Democratic Party and have joined Jung's campaign. Now that the election is approaching, they promise to increase housing supply and expedite projects, but there is a high likelihood they will revert to their old ways without reflection." Regarding electoral cooperation with the leadership of Jang Dong-hyuk, Oh stated, "We have entered a phase where strategic role division between the central party and candidates is necessary." He remarked, "With only about three weeks left until the election, it is now the candidates' time. The party's role is secondary." He added, "Elections are filled with the candidates' brands, policies, pledges, and messages. If the central party can assist, it should focus on the electoral landscape, such as actively fighting against the recent special prosecution law on canceled indictments and promoting its negative impacts." On the recent controversy over the 'national dividend,' Oh stated, "The central party needs to address whether the distribution related to semiconductors is a desirable government stance for the nation's future and articulate a vision for a better future as the opposition party."* This article has been translated by AI. 2026-05-14 20:25:09
Oh Se-hoon Proposes Debate with Jung Won-o Ahead of Local Elections Oh Se-hoon, the People Power Party candidate for Seoul mayor, reiterated his call for a one-on-one debate with Jung Won-o, the Democratic Party's candidate, on May 14, just six days before the local elections. Oh suggested that Democratic Party leader Jung Cheong-rae could moderate the debate, stating, "I hope at least one or two debates can be held." Speaking at a forum hosted by the Korean Newspaper and Broadcasting Editors Association in central Seoul, Oh said, "I am open to any format, time, place, or topic for the debate." He expressed frustration, saying, "Whenever I propose a debate, Jung gives evasive answers. Considering the public's right to know, I hope he reconsiders and shows at least a minimum level of courtesy." Oh emphasized the significance of the upcoming election, stating, "This election is a crossroads for whether the Lee Jae-myung administration will adopt a humble approach or transition into a more overt form of soft dictatorship. Absolute power corrupts absolutely; this is a historical truth." He warned that if Democratic Party candidates win in major local governments like Seoul and Busan, the current soft dictatorship under President Lee Jae-myung could become more severe. "I hope a minimum foothold for checking the government can be secured," he added. Oh also highlighted the choice between returning to the past of former Mayor Park Won-soon or moving toward the future. He pointed out that during Park's ten-year tenure, 389 redevelopment and reconstruction projects were canceled, leading to a significant reduction in housing supply and contributing to the current real estate crisis. He noted, "The people who implemented those policies are still within the Democratic Party and have joined Jung's campaign. Now that the election is approaching, they promise to increase housing supply and expedite projects, but there is a high likelihood they will revert to their old ways without reflection." Regarding electoral cooperation with the leadership of Jang Dong-hyuk, Oh stated, "We have entered a phase where strategic role division between the central party and candidates is necessary." He remarked, "With only about three weeks left until the election, it is now the candidates' time. The party's role is secondary." He added, "Elections are filled with the candidates' brands, policies, pledges, and messages. If the central party can assist, it should focus on the electoral landscape, such as actively fighting against the recent special prosecution law on canceled indictments and promoting its negative impacts." On the recent controversy over the 'national dividend,' Oh stated, "The central party needs to address whether the distribution related to semiconductors is a desirable government stance for the nation's future and articulate a vision for a better future as the opposition party."* This article has been translated by AI. 2026-05-14 20:25:09 -
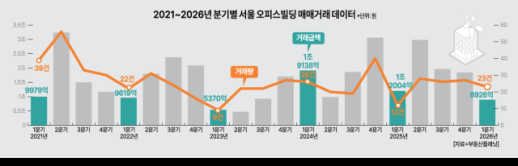 Seoul Office Transactions Plummet as Q1 Sales Halve In the first quarter of 2026, the Seoul office sales market experienced a significant contraction, with transaction amounts dropping by more than 50% compared to the previous quarter, alongside a decline in office sales. According to a report released on May 12 by Real Estate Planet, a commercial real estate platform, based on data from the Ministry of Land, Infrastructure and Transport, the volume of office building transactions in Seoul totaled 23 cases, with a transaction amount of 892.6 billion won in the first quarter of this year. This represents a 14.8% decrease in transaction volume and a 51.6% decrease in transaction amount compared to the previous quarter (27 cases, 1.8435 trillion won). However, compared to the same period last year (12 cases, 1.2004 trillion won), transaction volume increased by 91.7%, while transaction amount decreased by 25.6%. Regionally, the Gangnam and Seocho areas (GBD) maintained the same transaction volume as the previous quarter with 8 cases, while other regions recorded 12 cases. In contrast, the Yeongdeungpo and Mapo areas (YBD) dropped from 3 cases to 1, and the Jongno and Jung-gu areas (CBD) decreased from 4 cases to 2. Transaction amounts fell across all regions, with the CBD experiencing a dramatic decline from 942 billion won in the previous quarter to 64.5 billion won, a drop of 93.2%. The YBD saw a decrease to 16.5 billion won (down 62.2%), the GBD recorded 386.8 billion won (down 8.5%), and other regions totaled 424.8 billion won (down 2.4%). The office market also showed weakness, with the volume of office transactions in Seoul reaching 271 cases and a transaction amount of 499.2 billion won. This reflects a 20.3% decrease in transaction volume and a 25.8% decrease in transaction amount compared to the previous quarter (340 cases, 673.2 billion won). Compared to the same period last year, transaction volume increased by 0.4%, but transaction amount decreased by 11.6%. Corporations dominated both office building and office transactions. In the office building market, 15 out of 23 transactions were corporate purchases, accounting for 65.2%. In terms of transaction amount, corporate transactions totaled 766.4 billion won, representing 85.9% of the total. In the office market, corporate purchases were also the most prevalent, with 142 cases, while individual purchases were active with 126 cases, making up about 46.5% of the total. In terms of transaction amount, corporate transactions reached 421.9 billion won, accounting for 84.5% of the total. However, the REIT market continued to expand its office assets. As of March 2026, the total assets of REITs in the Seoul area amounted to 123.24 trillion won, a 4.2% increase from the previous month. Among these, office assets reached 43.45 trillion won, up 5.1% from the previous month and 32.8% from the same period last year. The supply of office facilities also showed an upward trend. According to building permit data from the Ministry of Land, Infrastructure and Transport, the total floor area of office facilities approved for use in the first quarter of this year was 221,476 square meters, a 159.3% increase from the previous quarter. The planned new supply of office facilities is also set to expand by 15.0% to 452,303 square meters compared to the previous quarter, particularly concentrated in Yeongdeungpo, Jongno, and Seongdong districts. The market contraction is also reflected in investment yield indicators. An additional review of the Real Estate Planet report revealed that the average investment yield for Seoul offices in the first quarter of this year was 2.35%, up from 1.84% in the previous quarter, although there were notable regional disparities. The investment yield in the CBD was 2.45%, in the GBD it was 2.65%, and in the YBD it was 2.20%, while other regions remained at 1.78%. In specific commercial areas, Gwanghwamun recorded a relatively high yield of 3.27%, and Gangnam-daero followed with 3.21%, while most other areas reported yields in the 1-2% range. Jung Soo-min, CEO of Real Estate Planet, stated, “In the first quarter of this year, the Seoul office sales market saw a decrease in large asset transactions, causing the transaction amount for office buildings to fall below 1 trillion won on a quarterly basis. As the expansion of office assets within REITs continues, along with an increase in the area of approved office facilities and a corporate-driven purchasing trend, the adjustment of price expectations between buyers and sellers and the resumption of large transactions in key regions will influence market trends in the second quarter.”* This article has been translated by AI. 2026-05-14 20:23:10
Seoul Office Transactions Plummet as Q1 Sales Halve In the first quarter of 2026, the Seoul office sales market experienced a significant contraction, with transaction amounts dropping by more than 50% compared to the previous quarter, alongside a decline in office sales. According to a report released on May 12 by Real Estate Planet, a commercial real estate platform, based on data from the Ministry of Land, Infrastructure and Transport, the volume of office building transactions in Seoul totaled 23 cases, with a transaction amount of 892.6 billion won in the first quarter of this year. This represents a 14.8% decrease in transaction volume and a 51.6% decrease in transaction amount compared to the previous quarter (27 cases, 1.8435 trillion won). However, compared to the same period last year (12 cases, 1.2004 trillion won), transaction volume increased by 91.7%, while transaction amount decreased by 25.6%. Regionally, the Gangnam and Seocho areas (GBD) maintained the same transaction volume as the previous quarter with 8 cases, while other regions recorded 12 cases. In contrast, the Yeongdeungpo and Mapo areas (YBD) dropped from 3 cases to 1, and the Jongno and Jung-gu areas (CBD) decreased from 4 cases to 2. Transaction amounts fell across all regions, with the CBD experiencing a dramatic decline from 942 billion won in the previous quarter to 64.5 billion won, a drop of 93.2%. The YBD saw a decrease to 16.5 billion won (down 62.2%), the GBD recorded 386.8 billion won (down 8.5%), and other regions totaled 424.8 billion won (down 2.4%). The office market also showed weakness, with the volume of office transactions in Seoul reaching 271 cases and a transaction amount of 499.2 billion won. This reflects a 20.3% decrease in transaction volume and a 25.8% decrease in transaction amount compared to the previous quarter (340 cases, 673.2 billion won). Compared to the same period last year, transaction volume increased by 0.4%, but transaction amount decreased by 11.6%. Corporations dominated both office building and office transactions. In the office building market, 15 out of 23 transactions were corporate purchases, accounting for 65.2%. In terms of transaction amount, corporate transactions totaled 766.4 billion won, representing 85.9% of the total. In the office market, corporate purchases were also the most prevalent, with 142 cases, while individual purchases were active with 126 cases, making up about 46.5% of the total. In terms of transaction amount, corporate transactions reached 421.9 billion won, accounting for 84.5% of the total. However, the REIT market continued to expand its office assets. As of March 2026, the total assets of REITs in the Seoul area amounted to 123.24 trillion won, a 4.2% increase from the previous month. Among these, office assets reached 43.45 trillion won, up 5.1% from the previous month and 32.8% from the same period last year. The supply of office facilities also showed an upward trend. According to building permit data from the Ministry of Land, Infrastructure and Transport, the total floor area of office facilities approved for use in the first quarter of this year was 221,476 square meters, a 159.3% increase from the previous quarter. The planned new supply of office facilities is also set to expand by 15.0% to 452,303 square meters compared to the previous quarter, particularly concentrated in Yeongdeungpo, Jongno, and Seongdong districts. The market contraction is also reflected in investment yield indicators. An additional review of the Real Estate Planet report revealed that the average investment yield for Seoul offices in the first quarter of this year was 2.35%, up from 1.84% in the previous quarter, although there were notable regional disparities. The investment yield in the CBD was 2.45%, in the GBD it was 2.65%, and in the YBD it was 2.20%, while other regions remained at 1.78%. In specific commercial areas, Gwanghwamun recorded a relatively high yield of 3.27%, and Gangnam-daero followed with 3.21%, while most other areas reported yields in the 1-2% range. Jung Soo-min, CEO of Real Estate Planet, stated, “In the first quarter of this year, the Seoul office sales market saw a decrease in large asset transactions, causing the transaction amount for office buildings to fall below 1 trillion won on a quarterly basis. As the expansion of office assets within REITs continues, along with an increase in the area of approved office facilities and a corporate-driven purchasing trend, the adjustment of price expectations between buyers and sellers and the resumption of large transactions in key regions will influence market trends in the second quarter.”* This article has been translated by AI. 2026-05-14 20:23:10 -
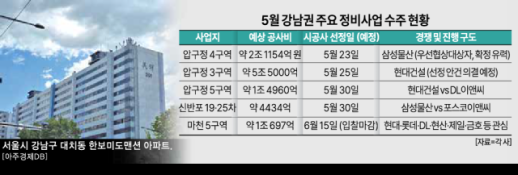 Major Reconstruction Projects in Gangnam Set to Begin in May The reconstruction market in Seoul's Gangnam district is entering a competitive phase this May. With the selection meetings for construction companies for Apgujeong districts 3, 4, and 5 scheduled throughout the month, major reconstruction projects in Shinbanpo and Songpa are also gaining momentum. Industry experts are viewing this month as a critical turning point for Gangnam reconstruction. According to the construction industry on May 12, selection meetings for construction companies are set to take place in Apgujeong and Shinbanpo, the two main battlegrounds for reconstruction in Gangnam. Specifically, districts 3 to 5 of the Apgujeong reconstruction project will finalize their construction companies this month. The six districts in Apgujeong are expected to accommodate between 11,000 and 12,000 households, with the construction costs for the three districts currently in the selection process exceeding 10 trillion won. The symbolic significance of the prime riverside location has intensified brand competition among construction firms. The first meeting will be held for Apgujeong district 4 on May 23, where Samsung C&T, selected as the preferred bidder last month, is likely to be confirmed as the construction company. On May 25, Apgujeong district 3 will hold a meeting to vote on selecting Hyundai Engineering & Construction as its contractor. This district, with a planned 4,121 units, is considered the largest single reconstruction site in Gangnam, with construction costs estimated at approximately 5.5 trillion won. The most intense competition is expected in Apgujeong district 5, where construction costs are around 1.496 trillion won. A meeting will take place on May 30 to decide between Hyundai Engineering & Construction and DL E&C. Both companies will hold a joint briefing on May 16, followed by a second briefing on the day of the meeting to sway the opinions of the members. Recently, competition has intensified over high-end brands, community features, and specialized designs, leading to rapidly increasing bidding conditions. According to the Gangnam District Office's housing construction project status, as of January, districts 2 to 5 in Apgujeong have all completed the establishment of their associations. Notably, Apgujeong district 3, which has faced delays since receiving its establishment approval in 2021, is expected to gain momentum with the upcoming selection process. There are expectations that the projects in the Apgujeong area, which have been stalled due to changes in maintenance plans and regulatory issues, will finally move forward following this bidding war. The selection meetings for the Shinbanpo districts 19 and 25 will also take place on May 30. Although this project involves a smaller scale of 614 units and construction costs of about 443.4 billion won compared to Apgujeong, its riverside location allows for a potential 49-story design, making it likely to become a landmark in the Seocho area. In this project, Samsung C&T and POSCO E&C will compete against each other. This marks the first time since January 2024 that the two companies have faced off at the same site, where POSCO E&C previously won a bid. The reconstruction fervor is also rapidly spreading to Songpa District. The redevelopment of the Machon 5 area has received its association establishment approval in January after 19 years of efforts. The association announced a bidding notice last month, with six construction firms, including Hyundai Engineering & Construction, Lotte Engineering & Construction, DL E&C, IPARK Hyundai Industrial Development, Jeil Construction, and Kumho Construction, attending the site briefing. The estimated construction cost is around 1.069 trillion won, with the bidding deadline set for June 15. This area is expected to emerge as a significant battleground following the major bids in Gangnam. In the Seongsu Strategic Maintenance Zone, Daewoo Construction and Lotte Construction have resumed their bidding processes, and the selection of a construction company for the Mokdong reconstruction site is also expected to gain traction in the second half of the year. Ko Jun-seok, a professor at Yonsei University's Sangnam Business School, stated, "The project conditions and design standards presented in the Apgujeong bidding war are likely to set benchmarks for future maintenance project bids."* This article has been translated by AI. 2026-05-14 20:20:14
Major Reconstruction Projects in Gangnam Set to Begin in May The reconstruction market in Seoul's Gangnam district is entering a competitive phase this May. With the selection meetings for construction companies for Apgujeong districts 3, 4, and 5 scheduled throughout the month, major reconstruction projects in Shinbanpo and Songpa are also gaining momentum. Industry experts are viewing this month as a critical turning point for Gangnam reconstruction. According to the construction industry on May 12, selection meetings for construction companies are set to take place in Apgujeong and Shinbanpo, the two main battlegrounds for reconstruction in Gangnam. Specifically, districts 3 to 5 of the Apgujeong reconstruction project will finalize their construction companies this month. The six districts in Apgujeong are expected to accommodate between 11,000 and 12,000 households, with the construction costs for the three districts currently in the selection process exceeding 10 trillion won. The symbolic significance of the prime riverside location has intensified brand competition among construction firms. The first meeting will be held for Apgujeong district 4 on May 23, where Samsung C&T, selected as the preferred bidder last month, is likely to be confirmed as the construction company. On May 25, Apgujeong district 3 will hold a meeting to vote on selecting Hyundai Engineering & Construction as its contractor. This district, with a planned 4,121 units, is considered the largest single reconstruction site in Gangnam, with construction costs estimated at approximately 5.5 trillion won. The most intense competition is expected in Apgujeong district 5, where construction costs are around 1.496 trillion won. A meeting will take place on May 30 to decide between Hyundai Engineering & Construction and DL E&C. Both companies will hold a joint briefing on May 16, followed by a second briefing on the day of the meeting to sway the opinions of the members. Recently, competition has intensified over high-end brands, community features, and specialized designs, leading to rapidly increasing bidding conditions. According to the Gangnam District Office's housing construction project status, as of January, districts 2 to 5 in Apgujeong have all completed the establishment of their associations. Notably, Apgujeong district 3, which has faced delays since receiving its establishment approval in 2021, is expected to gain momentum with the upcoming selection process. There are expectations that the projects in the Apgujeong area, which have been stalled due to changes in maintenance plans and regulatory issues, will finally move forward following this bidding war. The selection meetings for the Shinbanpo districts 19 and 25 will also take place on May 30. Although this project involves a smaller scale of 614 units and construction costs of about 443.4 billion won compared to Apgujeong, its riverside location allows for a potential 49-story design, making it likely to become a landmark in the Seocho area. In this project, Samsung C&T and POSCO E&C will compete against each other. This marks the first time since January 2024 that the two companies have faced off at the same site, where POSCO E&C previously won a bid. The reconstruction fervor is also rapidly spreading to Songpa District. The redevelopment of the Machon 5 area has received its association establishment approval in January after 19 years of efforts. The association announced a bidding notice last month, with six construction firms, including Hyundai Engineering & Construction, Lotte Engineering & Construction, DL E&C, IPARK Hyundai Industrial Development, Jeil Construction, and Kumho Construction, attending the site briefing. The estimated construction cost is around 1.069 trillion won, with the bidding deadline set for June 15. This area is expected to emerge as a significant battleground following the major bids in Gangnam. In the Seongsu Strategic Maintenance Zone, Daewoo Construction and Lotte Construction have resumed their bidding processes, and the selection of a construction company for the Mokdong reconstruction site is also expected to gain traction in the second half of the year. Ko Jun-seok, a professor at Yonsei University's Sangnam Business School, stated, "The project conditions and design standards presented in the Apgujeong bidding war are likely to set benchmarks for future maintenance project bids."* This article has been translated by AI. 2026-05-14 20:20:14 -
 Korea's Drug Safety Agency Official Advocates for Fast-Track Approval System to Boost K-Bio Global Competitiveness "To secure market leadership opportunities, a fast-track approval system is essential for domestic pharmaceutical and biotech companies," said Park Hyun-jung, head of the Bio-Pharmaceutical Approval Division at the Ministry of Food and Drug Safety (MFDS). She made these remarks on May 14 during the 16th Global Healthcare Forum (2026 GHF) held at the Korea Press Center in Seoul, organized by Aju Economic Daily. Park emphasized the necessity of establishing a fast-track approval system to facilitate the rapid growth of domestic pharmaceutical and biotech companies. The global pharmaceutical market is projected to expand to approximately $2.66 trillion by 2030. She noted that the export volume of domestic pharmaceuticals is also increasing rapidly, particularly in high-value sectors such as biopharmaceuticals and toxins. The export value rose from $7.04 billion in 2021 to a record $10.41 billion last year. The MFDS has been implementing the first phase of its "Drug Approval Review Innovation" initiative since January 2025. This initiative aims to enhance the speed and transparency of drug approvals, viewing expedited approvals as a foundation for global competitiveness in the pharmaceutical industry. Park pointed out that with many global blockbuster biopharmaceutical patents set to expire in the near future, new opportunities are emerging for the domestic biosimilar industry. She stressed the importance of increasing the speed and predictability of the approval process to secure market leadership. A key focus is on reducing the approval review period. The average time for new drug approvals has been cut from over 400 days to 295 days. "We operate dedicated review teams for each product and provide specialized consultations at each approval stage, including pre-review of supplementary submission materials," Park explained. "We have also prioritized the evaluation of Good Manufacturing Practices (GMP) and Good Clinical Practices (GCP) to reduce the burden on companies and enhance review efficiency." Following the first phase, the MFDS plans to implement a second phase of innovation. This will be discussed at the second core regulatory rationalization strategy meeting chaired by the President, focusing on fostering the biohealth industry and transitioning to a consumer-centered approval review system while significantly shortening the drug approval review period. The agency is gathering industry feedback on new drugs and biosimilars and plans to significantly increase the number of specialized review personnel. In April, the MFDS hired new staff for drug approval reviews and is pursuing additional recruitment. Park also introduced plans to simplify Phase III clinical trials for biosimilars. "With advancements in quality analysis technology and accumulated regulatory experience since the introduction of biosimilars, it has been confirmed that equivalence can be demonstrated for certain products without Phase III clinical trials," she said. She added, "We plan to demonstrate equivalence between new biopharmaceuticals and biosimilars primarily through quality comparisons and Phase I clinical trials, conducting Phase III trials only when sufficient evidence is lacking. We will also allow for pre-review requests of equivalence demonstration plans by the MFDS." Park concluded, "Through these regulatory improvements, patients will be able to access new drugs and biosimilars more quickly, and the industry will reduce its burden through shortened development and approval timelines. We also expect this to facilitate the early entry of domestic companies into the global market and expand exports."* This article has been translated by AI. 2026-05-14 20:18:00
Korea's Drug Safety Agency Official Advocates for Fast-Track Approval System to Boost K-Bio Global Competitiveness "To secure market leadership opportunities, a fast-track approval system is essential for domestic pharmaceutical and biotech companies," said Park Hyun-jung, head of the Bio-Pharmaceutical Approval Division at the Ministry of Food and Drug Safety (MFDS). She made these remarks on May 14 during the 16th Global Healthcare Forum (2026 GHF) held at the Korea Press Center in Seoul, organized by Aju Economic Daily. Park emphasized the necessity of establishing a fast-track approval system to facilitate the rapid growth of domestic pharmaceutical and biotech companies. The global pharmaceutical market is projected to expand to approximately $2.66 trillion by 2030. She noted that the export volume of domestic pharmaceuticals is also increasing rapidly, particularly in high-value sectors such as biopharmaceuticals and toxins. The export value rose from $7.04 billion in 2021 to a record $10.41 billion last year. The MFDS has been implementing the first phase of its "Drug Approval Review Innovation" initiative since January 2025. This initiative aims to enhance the speed and transparency of drug approvals, viewing expedited approvals as a foundation for global competitiveness in the pharmaceutical industry. Park pointed out that with many global blockbuster biopharmaceutical patents set to expire in the near future, new opportunities are emerging for the domestic biosimilar industry. She stressed the importance of increasing the speed and predictability of the approval process to secure market leadership. A key focus is on reducing the approval review period. The average time for new drug approvals has been cut from over 400 days to 295 days. "We operate dedicated review teams for each product and provide specialized consultations at each approval stage, including pre-review of supplementary submission materials," Park explained. "We have also prioritized the evaluation of Good Manufacturing Practices (GMP) and Good Clinical Practices (GCP) to reduce the burden on companies and enhance review efficiency." Following the first phase, the MFDS plans to implement a second phase of innovation. This will be discussed at the second core regulatory rationalization strategy meeting chaired by the President, focusing on fostering the biohealth industry and transitioning to a consumer-centered approval review system while significantly shortening the drug approval review period. The agency is gathering industry feedback on new drugs and biosimilars and plans to significantly increase the number of specialized review personnel. In April, the MFDS hired new staff for drug approval reviews and is pursuing additional recruitment. Park also introduced plans to simplify Phase III clinical trials for biosimilars. "With advancements in quality analysis technology and accumulated regulatory experience since the introduction of biosimilars, it has been confirmed that equivalence can be demonstrated for certain products without Phase III clinical trials," she said. She added, "We plan to demonstrate equivalence between new biopharmaceuticals and biosimilars primarily through quality comparisons and Phase I clinical trials, conducting Phase III trials only when sufficient evidence is lacking. We will also allow for pre-review requests of equivalence demonstration plans by the MFDS." Park concluded, "Through these regulatory improvements, patients will be able to access new drugs and biosimilars more quickly, and the industry will reduce its burden through shortened development and approval timelines. We also expect this to facilitate the early entry of domestic companies into the global market and expand exports."* This article has been translated by AI. 2026-05-14 20:18:00 -
 Emart Plans 500 Billion Won Capital Increase for Shinsegae Construction Emart is moving forward with a capital increase of 500 billion won for its subsidiary, Shinsegae Construction. According to a filing with the Financial Supervisory Service on May 14, Emart will participate in the capital increase through cash and asset contributions. The cash contribution will amount to 240 billion won (48%), while the asset contribution will be 260 billion won (52%). The assets being contributed include land and buildings owned by Emart at the Myungil branch. Emart will provide these assets in exchange for newly issued shares from Shinsegae Construction. Shinsegae Construction plans to issue 10 million common shares at a price of 50,000 won per share. Emart, as the major shareholder, will fully subscribe to the new shares. The payment deadline for the new shares is set for June 25. An Emart representative stated, "This decision aims to enhance financial structure and secure management stability through increased construction capital, thereby boosting market confidence and enabling stable business operations in the future." In related news, Emart reported a consolidated operating profit of 178.3 billion won for the first quarter, an 11.9% increase compared to the same period last year. Although net sales decreased by 1.3% to 7.1234 trillion won, profitability showed significant improvement. This marks the highest first-quarter operating profit since 2012. On a standalone basis, operating profit also rose by 9.7% to 146.3 billion won, achieving the highest first-quarter figure in eight years since 2018. Standalone total sales increased by 1.9% to 4.7152 trillion won.* This article has been translated by AI. 2026-05-14 20:13:58
Emart Plans 500 Billion Won Capital Increase for Shinsegae Construction Emart is moving forward with a capital increase of 500 billion won for its subsidiary, Shinsegae Construction. According to a filing with the Financial Supervisory Service on May 14, Emart will participate in the capital increase through cash and asset contributions. The cash contribution will amount to 240 billion won (48%), while the asset contribution will be 260 billion won (52%). The assets being contributed include land and buildings owned by Emart at the Myungil branch. Emart will provide these assets in exchange for newly issued shares from Shinsegae Construction. Shinsegae Construction plans to issue 10 million common shares at a price of 50,000 won per share. Emart, as the major shareholder, will fully subscribe to the new shares. The payment deadline for the new shares is set for June 25. An Emart representative stated, "This decision aims to enhance financial structure and secure management stability through increased construction capital, thereby boosting market confidence and enabling stable business operations in the future." In related news, Emart reported a consolidated operating profit of 178.3 billion won for the first quarter, an 11.9% increase compared to the same period last year. Although net sales decreased by 1.3% to 7.1234 trillion won, profitability showed significant improvement. This marks the highest first-quarter operating profit since 2012. On a standalone basis, operating profit also rose by 9.7% to 146.3 billion won, achieving the highest first-quarter figure in eight years since 2018. Standalone total sales increased by 1.9% to 4.7152 trillion won.* This article has been translated by AI. 2026-05-14 20:13:58 -
 Supreme Court Rules Semiconductor Purification System Technology is Industrial Technology The Supreme Court has determined that the technology for an eco-friendly ultra-pure water system used in semiconductor manufacturing qualifies as industrial technology. Therefore, leaking related materials constitutes a violation of the Industrial Technology Protection Act.On May 14, the Supreme Court's second division, led by Justice Kwon Young-jun, overturned a lower court's three-year prison sentence for an individual identified as A, who was charged with violating the Industrial Technology Protection Act and the Unfair Competition Prevention Act, as well as breach of trust.A, who was responsible for managing the construction of the ultra-pure water system at Samsung Engineering (now Samsung E&A), was accused of leaking files containing design templates that automatically calculate optimal parameters for the ultra-pure water system, control logic for automation, operational conditions, and interlock information. This was done in preparation for his move to a Chinese semiconductor consulting firm.Despite submitting a confidentiality agreement regarding the specifications and design documents of the ultra-pure water system, A allegedly retained these documents instead of returning or destroying them, leading to charges of violating the Unfair Competition Prevention Act. He was also accused of causing financial damage to Samsung Engineering by leaking these documents for personal gain.Both the first and second trials found A guilty of all charges except for the violation of the Industrial Technology Protection Act, sentencing him to three years in prison.The court stated regarding the Industrial Technology Protection Act violation, "The evidence submitted by the prosecution does not sufficiently establish that the design and construction technology for the ultra-pure water system used by the victim company falls within the scope of advanced technology as defined by the Ministry of Trade, Industry and Energy at the time of the incident."The Ministry's guidelines categorize advanced technologies and products, and the technology in question was claimed to be part of the 'plant engineering sector' under 'water resources (major category)/desalination (middle category)/membrane separation (sub-category)/optimal design technology for high-efficiency RO systems.' The key issue in the appeal was the interpretation of the middle category 'desalination.'The second trial explained, "The meaning of 'desalination' in the middle category refers to freshwater produced from seawater desalination, thus interpreting the 'optimal design technology for high-efficiency RO systems' as detailed technology related to seawater desalination. However, the technology in this case pertains to producing ultra-pure water for semiconductors from industrial water, which is unrelated to 'optimal design technology for high-efficiency RO systems' concerning seawater desalination."However, the Supreme Court found that the lower court erred in its judgment of not guilty regarding the Industrial Technology Protection Act, stating, "The lower court misunderstood the legal principles regarding the scope of 'technologies that fall under advanced technology' as defined by the old Industrial Technology Protection Act and the Industrial Development Act."The court further elaborated, "Considering the legislative purposes of the Industrial Development Act and the old Industrial Technology Protection Act, the general meanings and usages of 'freshwater' and 'desalination,' and the potential impact of this technology on the development, production, distribution, or use of products or services related to 'optimal design technology for high-efficiency RO systems,' the meaning of 'freshwater' in the middle category includes not only cases where the treated water's purpose is 'freshwater' but also cases where the source water is freshwater."* This article has been translated by AI. 2026-05-14 20:12:16
Supreme Court Rules Semiconductor Purification System Technology is Industrial Technology The Supreme Court has determined that the technology for an eco-friendly ultra-pure water system used in semiconductor manufacturing qualifies as industrial technology. Therefore, leaking related materials constitutes a violation of the Industrial Technology Protection Act.On May 14, the Supreme Court's second division, led by Justice Kwon Young-jun, overturned a lower court's three-year prison sentence for an individual identified as A, who was charged with violating the Industrial Technology Protection Act and the Unfair Competition Prevention Act, as well as breach of trust.A, who was responsible for managing the construction of the ultra-pure water system at Samsung Engineering (now Samsung E&A), was accused of leaking files containing design templates that automatically calculate optimal parameters for the ultra-pure water system, control logic for automation, operational conditions, and interlock information. This was done in preparation for his move to a Chinese semiconductor consulting firm.Despite submitting a confidentiality agreement regarding the specifications and design documents of the ultra-pure water system, A allegedly retained these documents instead of returning or destroying them, leading to charges of violating the Unfair Competition Prevention Act. He was also accused of causing financial damage to Samsung Engineering by leaking these documents for personal gain.Both the first and second trials found A guilty of all charges except for the violation of the Industrial Technology Protection Act, sentencing him to three years in prison.The court stated regarding the Industrial Technology Protection Act violation, "The evidence submitted by the prosecution does not sufficiently establish that the design and construction technology for the ultra-pure water system used by the victim company falls within the scope of advanced technology as defined by the Ministry of Trade, Industry and Energy at the time of the incident."The Ministry's guidelines categorize advanced technologies and products, and the technology in question was claimed to be part of the 'plant engineering sector' under 'water resources (major category)/desalination (middle category)/membrane separation (sub-category)/optimal design technology for high-efficiency RO systems.' The key issue in the appeal was the interpretation of the middle category 'desalination.'The second trial explained, "The meaning of 'desalination' in the middle category refers to freshwater produced from seawater desalination, thus interpreting the 'optimal design technology for high-efficiency RO systems' as detailed technology related to seawater desalination. However, the technology in this case pertains to producing ultra-pure water for semiconductors from industrial water, which is unrelated to 'optimal design technology for high-efficiency RO systems' concerning seawater desalination."However, the Supreme Court found that the lower court erred in its judgment of not guilty regarding the Industrial Technology Protection Act, stating, "The lower court misunderstood the legal principles regarding the scope of 'technologies that fall under advanced technology' as defined by the old Industrial Technology Protection Act and the Industrial Development Act."The court further elaborated, "Considering the legislative purposes of the Industrial Development Act and the old Industrial Technology Protection Act, the general meanings and usages of 'freshwater' and 'desalination,' and the potential impact of this technology on the development, production, distribution, or use of products or services related to 'optimal design technology for high-efficiency RO systems,' the meaning of 'freshwater' in the middle category includes not only cases where the treated water's purpose is 'freshwater' but also cases where the source water is freshwater."* This article has been translated by AI. 2026-05-14 20:12:16 -
 AI and Big Data Essential for Drug Development, Says Jeong Yoon-taek "High-quality data is essential for successful drug development," Jeong Yoon-taek, head of the Pharmaceutical Industry Strategy Research Institute, stated on May 14 at the 16th Global Healthcare Forum (2026 GHF) held at the Korea Press Center in Seoul. He emphasized the need for a transition to an AI-driven pharmaceutical industry. During his presentation, Jeong noted that the pharmaceutical and biotech sectors are entering the 'Pharma 5.0' era. He explained that the industry is facing challenges such as declining productivity in drug development and the expansion of next-generation modalities, making it difficult to maintain competitiveness with traditional development methods. "Without vast amounts of high-quality data, AI models cannot learn or predict effectively," he said, adding that the principle of 'capturing data to capture new drugs' is a key challenge in the era of AI drug development. Jeong highlighted the rapid changes in the drug development paradigm. In the past, candidate substances were identified primarily through researchers' experience and repetitive experiments. Now, the industry is shifting towards using extensive big data, including genomic, clinical, and literature data, with AI playing a crucial role in identifying candidate substances and optimizing clinical strategies. He also discussed the evolution of AI technology, which has progressed from rule-based systems to deep learning and generative AI, and is now advancing into autonomous AI and quantum technology integration. Previously, drug candidates were designed through computer-based virtual experiments, but now, laboratories are expanding to include robotic automation for actual substance synthesis and validation. Jeong stated that the increased use of AI is improving the efficiency of drug development. The introduction of AI-based predictive models and automation technologies is expected to shorten clinical trial durations, reduce development costs, and accelerate commercialization. "AI is not just speeding up research; it is transforming the entire drug development value chain," he said, noting that the use of AI is expanding from candidate discovery to preclinical, clinical, production, and distribution stages. He also pointed out the strategic shifts among global pharmaceutical giants. These companies are accelerating collaborations and mergers with AI-based platform firms, competing to build open ecosystems centered around data and platforms. He observed a shift from traditional vertically integrated structures to expertise-based network models in the industry. In contrast, Jeong criticized the limitations of the domestic AI drug development ecosystem. He noted that investments in South Korea are still heavily focused on early-stage candidate discovery, exacerbating the 'death valley' issue where foundational AI technologies fail to transition into medical practice. Many companies are struggling at the commercialization stage due to a lack of organic connections between funding, data, and validation systems. Jeong emphasized that the key to competing in AI drug development is ultimately data competitiveness. He stressed the importance of alleviating data monopolies among companies and establishing collaborative utilization systems. He cited the 'MELODI' case and collaborative learning models, stating, "We need to cultivate specialized talent, introduce standardized contract systems, and foster global partnerships. The government must also establish continuous support systems."* This article has been translated by AI. 2026-05-14 20:08:59
AI and Big Data Essential for Drug Development, Says Jeong Yoon-taek "High-quality data is essential for successful drug development," Jeong Yoon-taek, head of the Pharmaceutical Industry Strategy Research Institute, stated on May 14 at the 16th Global Healthcare Forum (2026 GHF) held at the Korea Press Center in Seoul. He emphasized the need for a transition to an AI-driven pharmaceutical industry. During his presentation, Jeong noted that the pharmaceutical and biotech sectors are entering the 'Pharma 5.0' era. He explained that the industry is facing challenges such as declining productivity in drug development and the expansion of next-generation modalities, making it difficult to maintain competitiveness with traditional development methods. "Without vast amounts of high-quality data, AI models cannot learn or predict effectively," he said, adding that the principle of 'capturing data to capture new drugs' is a key challenge in the era of AI drug development. Jeong highlighted the rapid changes in the drug development paradigm. In the past, candidate substances were identified primarily through researchers' experience and repetitive experiments. Now, the industry is shifting towards using extensive big data, including genomic, clinical, and literature data, with AI playing a crucial role in identifying candidate substances and optimizing clinical strategies. He also discussed the evolution of AI technology, which has progressed from rule-based systems to deep learning and generative AI, and is now advancing into autonomous AI and quantum technology integration. Previously, drug candidates were designed through computer-based virtual experiments, but now, laboratories are expanding to include robotic automation for actual substance synthesis and validation. Jeong stated that the increased use of AI is improving the efficiency of drug development. The introduction of AI-based predictive models and automation technologies is expected to shorten clinical trial durations, reduce development costs, and accelerate commercialization. "AI is not just speeding up research; it is transforming the entire drug development value chain," he said, noting that the use of AI is expanding from candidate discovery to preclinical, clinical, production, and distribution stages. He also pointed out the strategic shifts among global pharmaceutical giants. These companies are accelerating collaborations and mergers with AI-based platform firms, competing to build open ecosystems centered around data and platforms. He observed a shift from traditional vertically integrated structures to expertise-based network models in the industry. In contrast, Jeong criticized the limitations of the domestic AI drug development ecosystem. He noted that investments in South Korea are still heavily focused on early-stage candidate discovery, exacerbating the 'death valley' issue where foundational AI technologies fail to transition into medical practice. Many companies are struggling at the commercialization stage due to a lack of organic connections between funding, data, and validation systems. Jeong emphasized that the key to competing in AI drug development is ultimately data competitiveness. He stressed the importance of alleviating data monopolies among companies and establishing collaborative utilization systems. He cited the 'MELODI' case and collaborative learning models, stating, "We need to cultivate specialized talent, introduce standardized contract systems, and foster global partnerships. The government must also establish continuous support systems."* This article has been translated by AI. 2026-05-14 20:08:59 -
 Special Prosecutor Summons Kwak Jong-geun as First Suspect in Military Coup Investigation The second Comprehensive Special Investigation Team, led by Special Prosecutor Kwon Chang-young, summoned former Special Warfare Commander Kwak Jong-geun on May 14 as a suspect in connection with the state of emergency declared on December 3. This marks the first time the special prosecutor has summoned Kwak as a suspect.The special prosecutor's team is currently investigating Kwak regarding charges of rebellion under the Military Criminal Act, having called him in for questioning at 2 p.m. On March 3, Kwak appeared before the Comprehensive Special Investigation as a witness.Kwak is accused of sending subordinate soldiers to the National Assembly and the National Election Commission to incite riots during the declaration of the state of emergency.The special prosecutor has also implicated former President Yoon Suk Yeol, former Defense Minister Kim Yong-hyun, and former Director of the Defense Intelligence Command Noh Sang-won in rebellion charges. This is interpreted as the special prosecutor's view that deploying military personnel to state institutions such as the National Assembly constitutes rebellion against the state.However, it remains uncertain whether the special prosecutor can bring these charges to trial.Previously, the Prosecutor's Special Investigation Unit had considered applying rebellion charges against former President Yoon but ultimately decided against it, citing difficulties in prosecution. It was reported that the rationale was that 'it is difficult to apply rebellion charges since the incident involved Yoon, the military commander, as the leader of the insurrection.'Both Yoon and Kim have refused to comply with the special prosecutor's summons, arguing that it constitutes a double investigation of the same case for which they are already facing insurrection charges.* This article has been translated by AI. 2026-05-14 20:07:33
Special Prosecutor Summons Kwak Jong-geun as First Suspect in Military Coup Investigation The second Comprehensive Special Investigation Team, led by Special Prosecutor Kwon Chang-young, summoned former Special Warfare Commander Kwak Jong-geun on May 14 as a suspect in connection with the state of emergency declared on December 3. This marks the first time the special prosecutor has summoned Kwak as a suspect.The special prosecutor's team is currently investigating Kwak regarding charges of rebellion under the Military Criminal Act, having called him in for questioning at 2 p.m. On March 3, Kwak appeared before the Comprehensive Special Investigation as a witness.Kwak is accused of sending subordinate soldiers to the National Assembly and the National Election Commission to incite riots during the declaration of the state of emergency.The special prosecutor has also implicated former President Yoon Suk Yeol, former Defense Minister Kim Yong-hyun, and former Director of the Defense Intelligence Command Noh Sang-won in rebellion charges. This is interpreted as the special prosecutor's view that deploying military personnel to state institutions such as the National Assembly constitutes rebellion against the state.However, it remains uncertain whether the special prosecutor can bring these charges to trial.Previously, the Prosecutor's Special Investigation Unit had considered applying rebellion charges against former President Yoon but ultimately decided against it, citing difficulties in prosecution. It was reported that the rationale was that 'it is difficult to apply rebellion charges since the incident involved Yoon, the military commander, as the leader of the insurrection.'Both Yoon and Kim have refused to comply with the special prosecutor's summons, arguing that it constitutes a double investigation of the same case for which they are already facing insurrection charges.* This article has been translated by AI. 2026-05-14 20:07:33 -
 Pharos AI Bio CEO Nam Ki-yeob: AI Era in Drug Development Enhances K-New Drug Success Rates Artificial intelligence (AI) is transforming drug development from a race for speed to a competition for success rates. The technology has evolved to not only identify candidate substances more quickly but also to filter out toxic risks in advance and derive optimal drug candidates. South Korea's AI drug development company, Pharos AI Bio, is accelerating the development of treatments for rare and intractable diseases and expanding into next-generation modalities with its proprietary platform.Nam Ki-yeob, CEO of Pharos AI Bio, explained at the 16th Global Healthcare Forum (2026 GHF) held on May 14 at the Korea Press Center in Seoul, that "AI drug development has entered the era of 'agent AI,' where it can now formulate hypotheses and make decisions after going through computer-based drug design, machine learning, and deep learning stages."Major domestic bio companies are actively responding to this global change based on their own platforms in the AI era. Pharos AI Bio's core competitive advantage lies in its self-developed AI platform, Chemiverse.Chemiverse is a platform that supports the entire drug development cycle by combining proteomics AI with generative model-based candidate substance design technology, based on multi-omics big data. It addresses the inefficiencies and high costs associated with drug development, covering everything from target discovery to candidate substance design, lead optimization, and the establishment of preclinical and clinical strategies.Nam stated, "The success rate of our pipeline using the AI platform from Phase 1 to Phase 2 clinical trials reaches 87.5%." This reflects a reduction in the high failure rate, a fundamental challenge in drug development, through data and predictive technology. He added, "By combining a strategy focused on rare and intractable diseases with biomarker-based patient selection and open innovation, we are enhancing the likelihood of success in drug development."A notable example of the company's AI application is the acute myeloid leukemia (AML) treatment, Rasmotinib (PHI-101). This candidate substance is a FLT3 inhibitor developed for patients who do not respond to or are resistant to existing treatments. Nam remarked, "Rasmotinib has achieved a 50% complete response rate in global Phase 1 clinical trials, emerging as a new hope for patients resistant to standard therapies." He added that they are pushing for early commercialization through entry into Phase 2 trials, conditional sales approval, and technology transfers.Additionally, the company plans to expand into next-generation modalities such as antibody-drug conjugates (ADCs) and targeted protein degraders (TPDs) based on Chemiverse. They aim to broaden the scope of drug development by combining structure-based chemical design capabilities with specialized experience in rare and intractable targets and core structural big data.Nam emphasized the importance of building competitiveness through in-house drug development, stating, "AI has evolved from a simple tool to an intelligent partner that supports decision-making throughout the entire drug development process." He further stressed that in the AI era, K-bio's success will depend not just on identifying good drug candidates but also on how well data is accumulated and interpreted to connect with the global value chain. A full value chain strategy that supports technology validation, capital acquisition, and global expansion is essential for the competitiveness of K-new drugs to become a reality.Finally, Nam reiterated, "Pharos AI Bio will continue to strengthen its research and development competitiveness in innovative drugs for rare and intractable diseases and in various modality areas such as ADCs and TPDs based on Chemiverse."* This article has been translated by AI. 2026-05-14 20:05:28
Pharos AI Bio CEO Nam Ki-yeob: AI Era in Drug Development Enhances K-New Drug Success Rates Artificial intelligence (AI) is transforming drug development from a race for speed to a competition for success rates. The technology has evolved to not only identify candidate substances more quickly but also to filter out toxic risks in advance and derive optimal drug candidates. South Korea's AI drug development company, Pharos AI Bio, is accelerating the development of treatments for rare and intractable diseases and expanding into next-generation modalities with its proprietary platform.Nam Ki-yeob, CEO of Pharos AI Bio, explained at the 16th Global Healthcare Forum (2026 GHF) held on May 14 at the Korea Press Center in Seoul, that "AI drug development has entered the era of 'agent AI,' where it can now formulate hypotheses and make decisions after going through computer-based drug design, machine learning, and deep learning stages."Major domestic bio companies are actively responding to this global change based on their own platforms in the AI era. Pharos AI Bio's core competitive advantage lies in its self-developed AI platform, Chemiverse.Chemiverse is a platform that supports the entire drug development cycle by combining proteomics AI with generative model-based candidate substance design technology, based on multi-omics big data. It addresses the inefficiencies and high costs associated with drug development, covering everything from target discovery to candidate substance design, lead optimization, and the establishment of preclinical and clinical strategies.Nam stated, "The success rate of our pipeline using the AI platform from Phase 1 to Phase 2 clinical trials reaches 87.5%." This reflects a reduction in the high failure rate, a fundamental challenge in drug development, through data and predictive technology. He added, "By combining a strategy focused on rare and intractable diseases with biomarker-based patient selection and open innovation, we are enhancing the likelihood of success in drug development."A notable example of the company's AI application is the acute myeloid leukemia (AML) treatment, Rasmotinib (PHI-101). This candidate substance is a FLT3 inhibitor developed for patients who do not respond to or are resistant to existing treatments. Nam remarked, "Rasmotinib has achieved a 50% complete response rate in global Phase 1 clinical trials, emerging as a new hope for patients resistant to standard therapies." He added that they are pushing for early commercialization through entry into Phase 2 trials, conditional sales approval, and technology transfers.Additionally, the company plans to expand into next-generation modalities such as antibody-drug conjugates (ADCs) and targeted protein degraders (TPDs) based on Chemiverse. They aim to broaden the scope of drug development by combining structure-based chemical design capabilities with specialized experience in rare and intractable targets and core structural big data.Nam emphasized the importance of building competitiveness through in-house drug development, stating, "AI has evolved from a simple tool to an intelligent partner that supports decision-making throughout the entire drug development process." He further stressed that in the AI era, K-bio's success will depend not just on identifying good drug candidates but also on how well data is accumulated and interpreted to connect with the global value chain. A full value chain strategy that supports technology validation, capital acquisition, and global expansion is essential for the competitiveness of K-new drugs to become a reality.Finally, Nam reiterated, "Pharos AI Bio will continue to strengthen its research and development competitiveness in innovative drugs for rare and intractable diseases and in various modality areas such as ADCs and TPDs based on Chemiverse."* This article has been translated by AI. 2026-05-14 20:05:28
