South Korea’s biopharmaceutical exports posted a record first-quarter total despite prolonged Middle East risks, led by contract development and manufacturing (CDMO) firms, regulators said. At the same time, domestically focused drugmakers are moving to emergency measures as uncertainty grows over supplies of packaging-related materials.
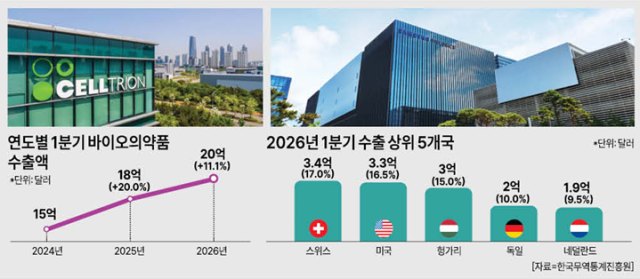
The Ministry of Food and Drug Safety said April 12 that biopharmaceutical exports in the first quarter totaled $2.0 billion (about 2.96 trillion won), up 11.1% from a year earlier. Biopharma accounted for 71% of Korea’s total pharmaceutical exports of $2.8 billion. The quarterly total extended a three-year rise from $1.5 billion in 2024 to $1.8 billion last year and $2.0 billion this year, underscoring the competitiveness of Korea’s CDMO industry.
Monthly exports were steady: $660 million in January, up 11.9% year over year; $690 million in February, up 25.4%; and $650 million in March, up 2%. By destination, Switzerland took 17%, the United States 16.5%, Hungary 15%, Germany 10% and the Netherlands 9.5%. The top five markets accounted for 68.4% of the total.
Industry officials attributed the expansion in Europe to a combination of co-development with global drugmakers, technology exports and favorable perceptions of biosimilars. “As CDMO competitiveness and production infrastructure reach global standards, companies are actively building direct production and supply systems for Europe,” an industry official said.
Samsung Biologics and Celltrion, among large companies with in-house production and CDMO capabilities, have helped drive the boom. Lotte Biologics, a late entrant to the CDMO market, is expanding the business with more than 1 trillion won in group-level investment.
The government is also stepping up support for global expansion. A special law enacted last year to provide regulatory support for biopharmaceutical CDMO companies is set to take effect in December. Key provisions include streamlining production licensing and cutting the number of documents required for GMP evaluation submissions to four from 11.
The law is intended to provide a framework allowing export-oriented CDMO companies to enter overseas markets without obtaining a separate manufacturing license. The ministry is also running a pilot program to certify manufacturing sites for raw materials and providing global regulatory information, while supporting the industry’s push to build a “fast-track review system.”
While exports remain strong, Middle East-related risks are weighing on the broader domestic medicine supply chain. As fighting between the United States and Iran drags on, the Strait of Hormuz has remained blocked, and international oil prices at one point topped $100 a barrel. With crude supply uncertainty spreading to refiners, naphtha prices have recently jumped by 20% to 30%.
Naphtha is a key feedstock for medical plastics such as IV fluid bags and packaging containers. That has heightened concerns among suppliers of IV solutions, including JW Pharmaceutical and HK inno.N. The pharmaceutical industry is also working to prevent stockpiling by hospitals, clinics and pharmacies.
Yuhan Corp. has tightened inventory management for its acetaminophen in IV-bag form. HK inno.N, an IV-solution supplier, has adjusted some excessive orders to block stockpiling in advance.
The ministry recently held an emergency meeting with major drugmakers to discuss short-term steps including using alternative materials, adjusting shipments and freezing inventories. Major companies are expanding stockpiles and diversifying suppliers amid concerns over shortages of key inputs such as naphtha, while the government is strengthening supply-chain monitoring and seeking joint measures with the industry.
* This article has been translated by AI.
Copyright ⓒ Aju Press All rights reserved.

