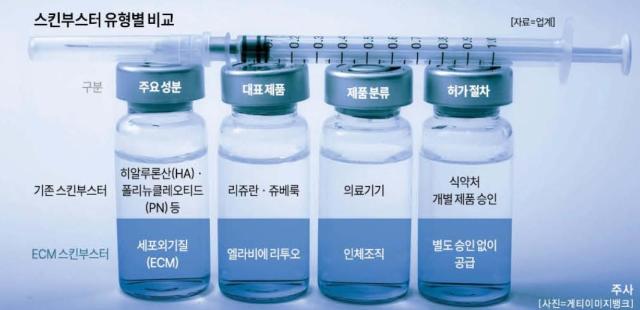
Competition in South Korea’s skin booster market is shifting quickly as more pharmaceutical and biotech companies enter the field and products based on human tissue spread. Skin boosters refer to injectable procedures that deliver substances such as hyaluronic acid and collagen into the dermis to help moisturize, improve elasticity and support skin regeneration. The market, long led by products such as PharmaResearch’s Rejuran and Juvlook, is increasingly seeing late challengers promote ECM (extracellular matrix) offerings.
According to Grand View Research, the domestic skin booster market totaled $87.7 million (about 128.7 billion won) in 2024 and is projected to grow 16% annually through 2030.
Rejuran has dominated the sector, and PharmaResearch’s revenue topped 500 billion won last year on growth in its aesthetics business centered on the product. But as the market expands and new products multiply, its once-dominant position is weakening, with ECM skin boosters at the center of the change.
L&C Bio’s “Ellavie Rituo,” launched in late 2024, is made by leaving only the collagen structure from donated deceased skin tissue, turning it into powder and injecting it. The company now has about 2,000 hospital and clinic accounts.
L&C Bio is also reviewing an additional production-facility expansion, targeting October. If carried out, output is expected to rise from 30,000 units to as many as more than 150,000 units a month by the end of this year. Lee Seung-eun, an analyst at Yuanta Securities, forecast that Rituo revenue could expand to 50 billion won this year from about 6 billion won a year earlier.
Other drug and biotech companies are also moving in. CGBio’s affiliate CGBiomeditech, which is expanding its ECM business based on its parent company’s human-tissue processing capabilities, recently acquired CGBio’s Seongnam human-tissue processing tissue bank to secure a production base.
GC Wellbeing has launched “GCell Rebonne” and is accelerating its push into medical aesthetics. MS Bio handles tissue processing, while GC Wellbeing has built a system to manage and supply raw materials through a distribution tissue bank at its Eumseong plant in North Chungcheong Province. Hugel has also joined the market by signing a domestic distribution contract for Hans Biomed’s “CellrDM.”
Differences in oversight are also fueling the spread of ECM products. Rituo is classified as human tissue, meaning it is subject to procedures different from medical devices such as Rejuran and Juvlook. Medical devices must go through the Ministry of Food and Drug Safety’s process for each product, including intended use and indications. Human tissue, by contrast, can be supplied to hospitals and clinics without separate approval for each product, creating a relatively lower barrier to entry. Lee Jun-seok, an analyst at Hanyang Securities, said, “The ECM skin booster business depends on stable procurement of raw materials,” adding that success will hinge on “how reliably companies can secure human tissue that meets regulatory requirements.”
At the same time, debate is intensifying over the use of human tissue for cosmetic purposes. Critics have raised ethical concerns because donated tissue is used in commercial procedures. Kwon Dong-ju, an attorney at law firm Yulchon, said at the K-Bio Health Forum on April 16, hosted by Democratic Party lawmaker Seo Dong-seok under the theme “Reasonable regulatory measures for the proper use of donated human tissue,” that it is not reasonable for products with the same purpose and method of administration as other injectable skin boosters already approved as medical devices to be regulated entirely differently. He argued that the Human Tissue Act should explicitly ban cosmetic use.
* This article has been translated by AI.
Copyright ⓒ Aju Press All rights reserved.