Hyundai Bioscience said April 20 that South Korea’s Ministry of Food and Drug Safety has given final approval to a revised investigational new drug plan for a combination-therapy clinical trial in prostate cancer using its broad-based “environment normalization” agent, Penetrium.
The company said it will submit related documents on April 21 to the Institutional Review Board at Seoul National University Hospital, then hold a site initiation visit in mid-May before starting patient enrollment and dosing.
The trial is examining whether reduced drug response can stem not only from cancer-cell mutations but also from a physical barrier in the tumor microenvironment that can create what it calls “pseudo-resistance,” in which drugs fail to reach the tumor and the lack of response is mistaken for tumor evolution.
The newly approved change adds an AR-V7 genetic-variant test at the patient screening stage. Patients with “true resistance” (AR-V7 positive), in which the receptor itself is altered and the drug does not work, will be excluded. The study will instead select patients with “pseudo-resistance” (AR-V7 negative) and treat them with a combination of enzalutamide and Penetrium.
The company said that if the trial produces successful interim results, prostate cancer patients could potentially continue benefiting from existing targeted therapies without moving to chemotherapy that can involve severe side effects.

Huons Group said April 20 it held a donation ceremony after joining the Gyeonggi branch of the Community Chest of Korea’s “Sharing Prestigious Company” program.
Attendees included Song Soo-young, CEO of Huons Global and Huons; Kang Min-jong, CEO of Humedix; and Kwon In-wook, chairman of the Gyeonggi Community Chest of Korea, the company said.
The program is a network of corporate donors that have contributed at least 100 million won. Huons Group said three of its companies decided to join together to continue donations, following ongoing community support, medical volunteer work and scholarship programs.
The donations will be used through the Gyeonggi Community Chest of Korea to support living and medical expenses for low-income households and to improve educational environments, the company said.
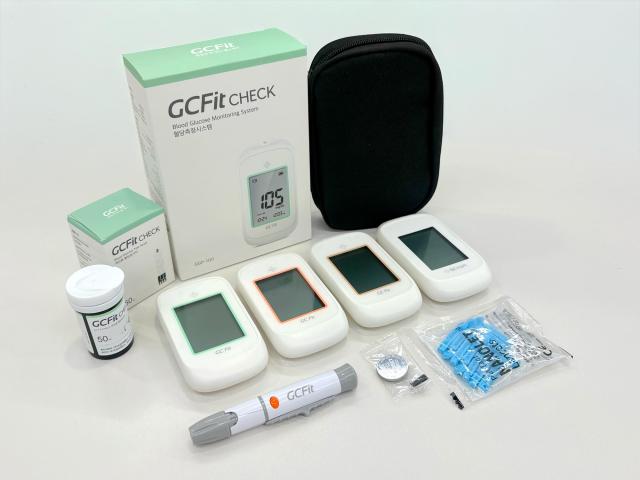
GC Green Cross MS said April 20 that its new blood glucose monitoring system, the GC Fit (model GGP-100), has received certification under the European Union’s In Vitro Diagnostic Medical Devices Regulation, known as CE-IVDR.
CE-IVDR tightens performance and safety requirements compared with the previous CE-IVDD framework and is required for entry into the EU market, the company said.
GC Green Cross MS said the GGP-100 met the strengthened standards despite being classified as a high-risk Class C product, demonstrating safety and performance. The company said the certification also secures EU-level performance and safety coverage for its full lineup of blood glucose meters, including existing products.
The company said the approval enables EU market entry about two months earlier than planned. It also said it plans to expand exports by obtaining CE-IVDR certification for an optical hemoglobin measurement product.

Jaseng Medical Foundation said April 20 it held a handover ceremony to provide soccer equipment for Bucheon FC 1995 youth teams.
The foundation said the support is intended to help create a stable training environment for young players aspiring to become national team members. About 100 players will receive uniforms, soccer cleats and balls, among other items, worth about 5 million won. Jaseng Healthcare also sponsored health functional foods, it said.
The foundation said it plans to continue support under the aim of encouraging the youth teams’ efforts, while monitoring training conditions and considering additional measures such as medical support.
Bucheon Jaseng Korean Medicine Hospital has provided medical support to Bucheon FC 1995 under a partnership spanning 17 years, and has participated as the club’s main sponsor since last year.
* This article has been translated by AI.
Copyright ⓒ Aju Press All rights reserved.