SEOUL, April 22 (AJP) - A research team led by Professor Kim Ju-mee at Sookmyung Women's University's Division of Pharmacy has identified a new pathological mechanism for GNE myopathy, a rare muscle disorder, and proposed a potential treatment strategy. By analyzing the genetic pathways of the disease, the team discovered that an existing drug approved by the U.S. Food and Drug Administration could be repurposed to restore cellular functions that are impaired by the condition, the university said Wednesday.
UDP-GlcNAc 2-epimerase/N-acetylmannosamine kinase (GNE) myopathy is a rare genetic disease caused by mutations in the GNE gene, which leads to a decrease in the synthesis of sialic acid. This deficiency results in progressive muscle weakness throughout the body, and the condition currently lacks a fundamental cure.
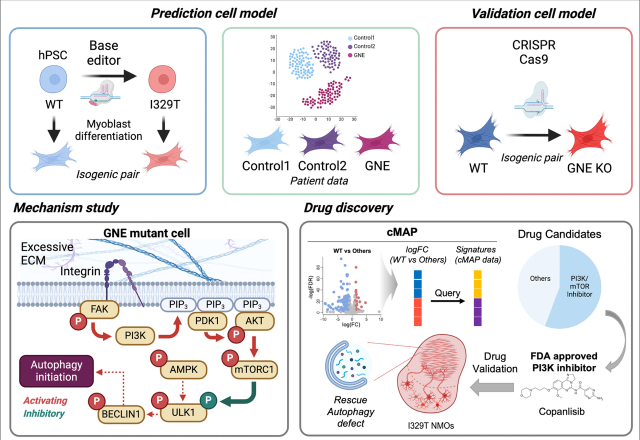
The research team used human pluripotent stem cell-derived muscle models to conduct a transcriptome analysis. Through this process, they confirmed that dysfunction in autophagy—the body's natural process of cleaning out damaged cells—is a primary driver of the disease's progression.
Specifically, the study revealed that an excessive production of the extracellular matrix abnormally activates the PI3K-AKT-mTORC1 signaling pathway. This activation inhibits ULK1, a factor necessary for initiating autophagy, effectively blocking the cell's ability to maintain itself.
Using a drug screening technique based on these genetic findings, the researchers identified Copanlisib as a strong candidate for treatment. Copanlisib, which is already an FDA-approved PI3K inhibitor, was found to suppress the abnormal signaling and restore ULK1 activity, thereby normalizing the autophagy process in the diseased cells.
These results were further validated using human stem cell-derived neuromuscular organoid models. The study was conducted as a joint effort with a research team led by Professor Cha Hyuk-jin from Seoul National University (SNU).

"This research is significant because it goes beyond identifying the fundamental molecular mechanism of the disease to suggest a treatment strategy using an already approved drug," Professor Kim Ju-mee said. "In particular, the use of drug repositioning for rare diseases greatly increases the possibility of clinical application."
The findings were published in the April 2026 issue of Experimental & Molecular Medicine, a leading international journal in the field of molecular medicine.
[Reference Information]
◆Experimental & Molecular Medicine (Impact Factor 12.9, JCR top 4.1%)
◆Title: Defective autophagy in GNE myopathy is rescued by inhibition of noncanonical Akt–mTORC1 activation across multiple isogenic models
◆Link/DOI: https://bit.ly/4cXXoOP
Copyright ⓒ Aju Press All rights reserved.


![[Coronavirus] Research team warns of COVID-19-related inflammation of heart muscle](https://image.ajunews.com/content/image/2020/04/17/20200417161231134110_278_163.jpg)
